-

30+
Years of clinical data services
-

50%
Reduction in TFL development time
-

30%
Cost reduction through automation and open source solutions.
-

250+
Clinical trials supported
-

30+
NDA, MAA and PMDA submissions supported
What we do
Unlocking The Power of Data to Drive Breakthroughs in Clinical Research
From data analysis and visualization to open source solutions, Clymb Clinical offers a diverse set of services designed to help clients harness the power of their data.
-

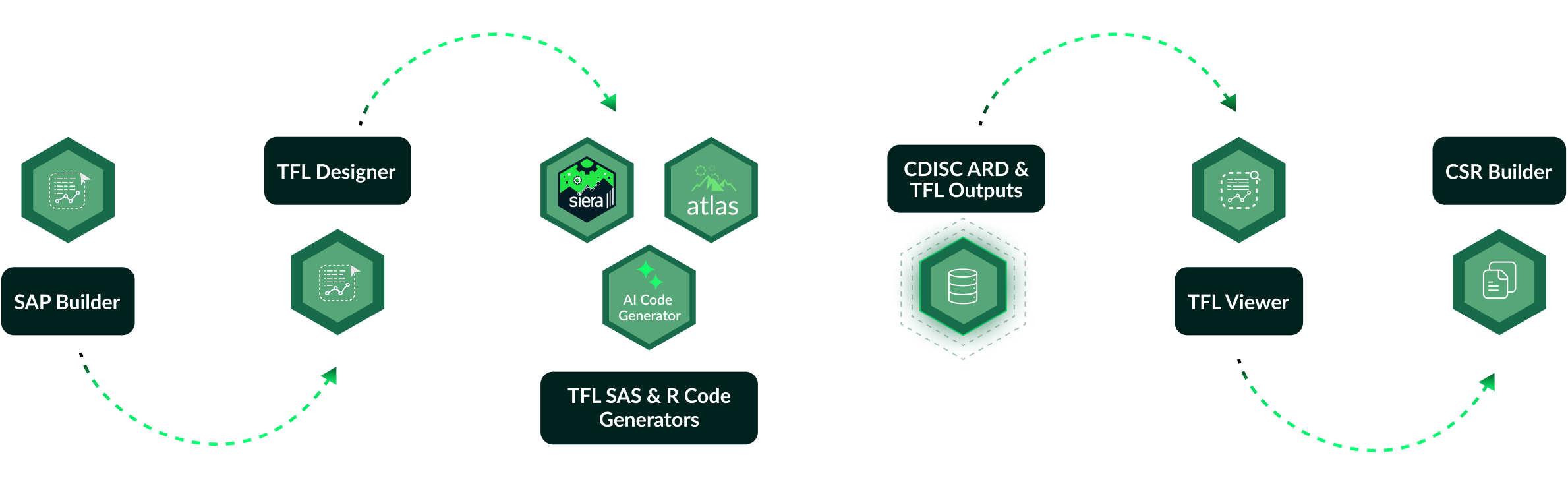
Clymbr Hub
The Clymbr Hub brings together Clymb's full suite of automation and AI solutions, including TFL Designer, TFL Code Generators, and TFL Viewer, into a single integrated workspace.
-

Submify
Submify, a dedicated Data Submission team of Clymb Clinical, created to address the unique needs of small and mid-sized sponsor companies.
-

Outsourcing Services
Clymb Clinical offers a comprehensive range of outsourcing services to support your clinical trials, from expert protocol design to advanced statistical programming and regulatory submission support.
-

Functional Services
Our FSP model is a flexible outsourcing approach that allows companies to leverage the expertise of external service providers to support specific functions or tasks within their clinical trial programs.
Got Questions? We've Got Answers
What is TFL Designer and what are the benefits of using such a solution?
TFL Designer, or Tables, Figures, and Listings Designer, is a specialized tool developed by Clymb Clinical to streamline the creation of TFLs in clinical trial data analysis. It digitizes and automates the design of these essential components, offering machine-readable analysis results metadata. The benefits of using TFL Designer include improved efficiency in TFL creation, customization flexibility, alignment with CDISC Analysis Results Standards, and support for downstream automation processes including generation of Analysis Results Data and TFL codes/outputs. This robust solution streamlines data analysis workflows (keeping end in mind - going from study objectives to data ection i.e. TFL → ADaM → SDTM → CRF), leading to time savings, enhanced data quality, and more effective decision-making.
In addition to these advantages, we are actively working on a community free version. This initiative will empower the industry with broader access to TFL Designer, fostering collaboration and knowledge sharing among stakeholders. Users of the community version will benefit from a growing library of templates, use cases, and standards, accelerating TFL design and automation. Furthermore, the incorporation of FDA templates, based on recent FDA Safety Tables and Figures - Integrated Guide (STF-IG), in the community version enhances its utility and aligns it with the latest regulatory requirements. By offering a community version, we aim to contribute to a more efficient and standardized approach to clinical trial data analysis across the industry.
Where can I find more info on the TFL Designer and next gen tools?
Thank you for your interest in our TFL Designer and next gen tools. We will be regularly updating our website with more information on these solutions, so please visit our website for the latest updates. At Clymb, we offer a comprehensive range of services and solutions to meet the evolving needs of the clinical research industry, including cutting-edge software tools and expert consulting services. We are committed to providing our clients with the best possible solutions, and we look forward to helping you achieve your clinical research goals.
How does Clymb use automation in their service offerings?
At Clymb Clinical, we are committed to leveraging the latest technologies and automation tools to deliver fast, efficient, and high-quality clinical trial services to our clients. We use automation in a variety of ways, from data mapping and analysis to TFL generation and regulatory submission support. By automating routine tasks and processes, we can reduce the risk of errors and increase efficiency, allowing our team to focus on more complex and strategic tasks.
What are the advantages to using Clymb over other service providers?
Clymb Clinical offers a unique combination of expertise, experience, and innovation that sets us apart from other service providers in the clinical research industry. With over 30 years of experience in clinical data management and biostatistics, we have a deep understanding of the challenges and opportunities facing our clients. Our commitment to innovation and automation ensures that we can deliver fast, efficient, and high-quality services that meet the evolving needs of the industry. And our team of experts is dedicated to providing personalized service and support that exceeds our clients' expectations.

You have different questions?
Dont worry! Our team will answer them for you