CSR Builder
Automate Clinical Study Report Development With Precision and Speed
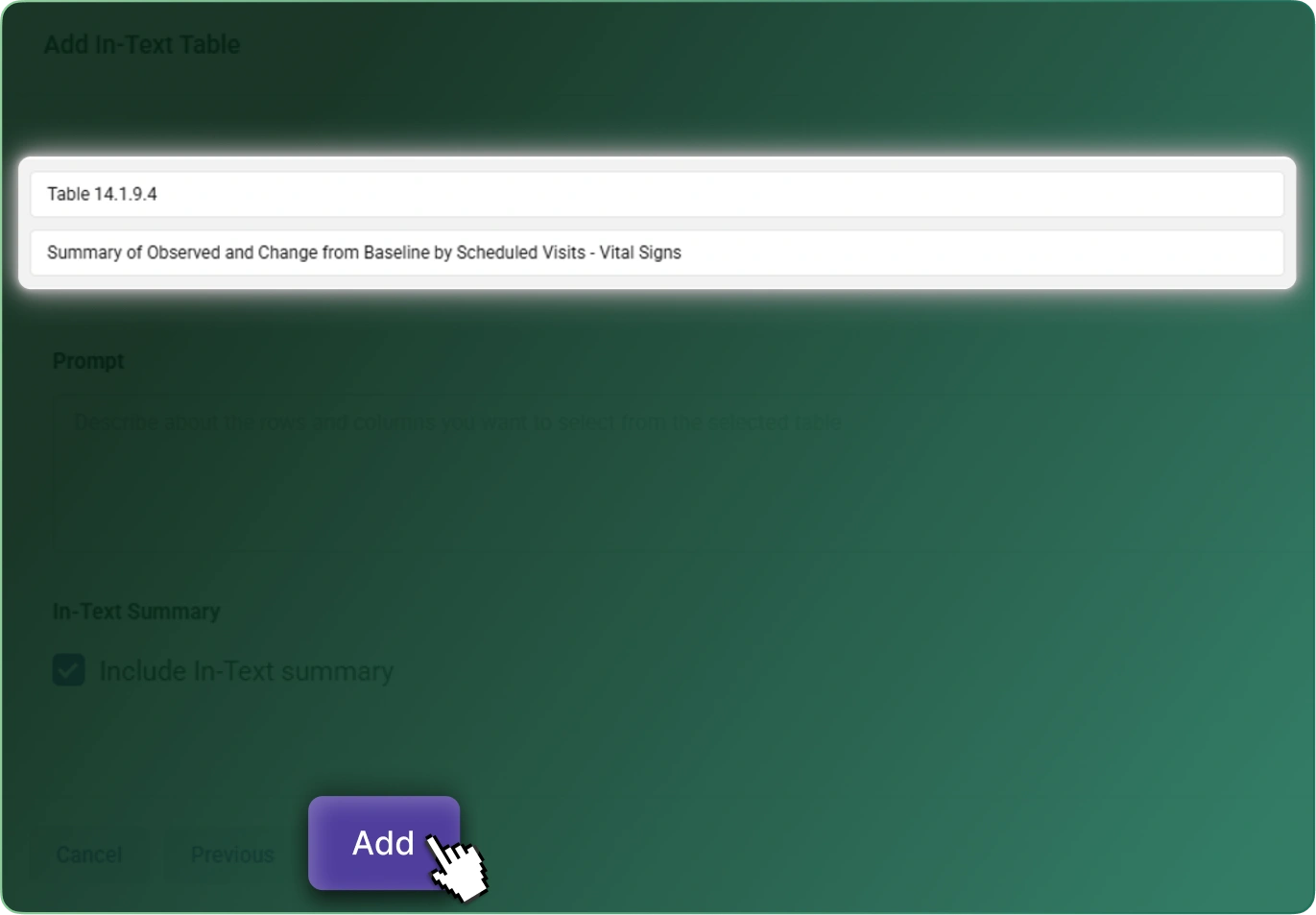
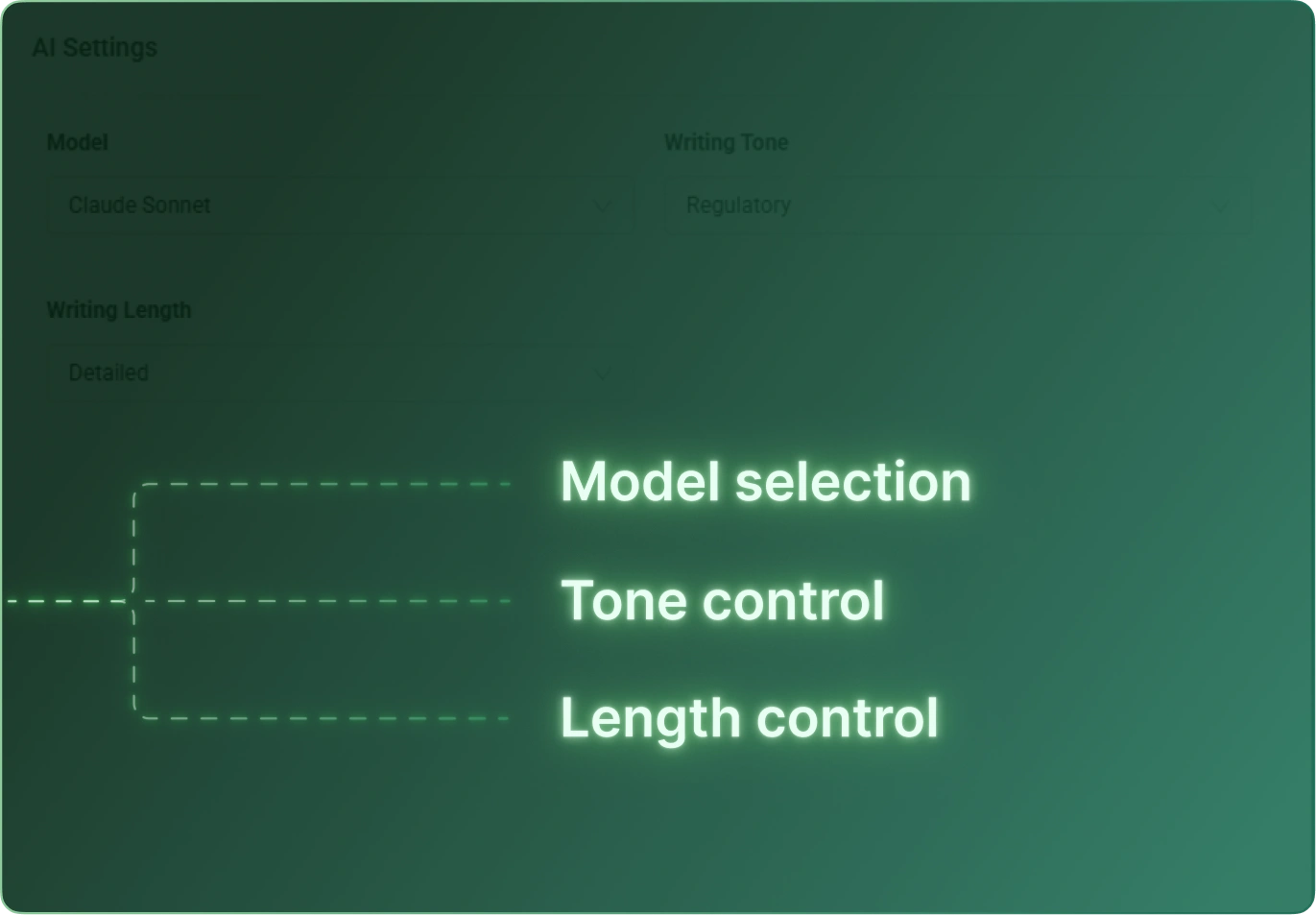
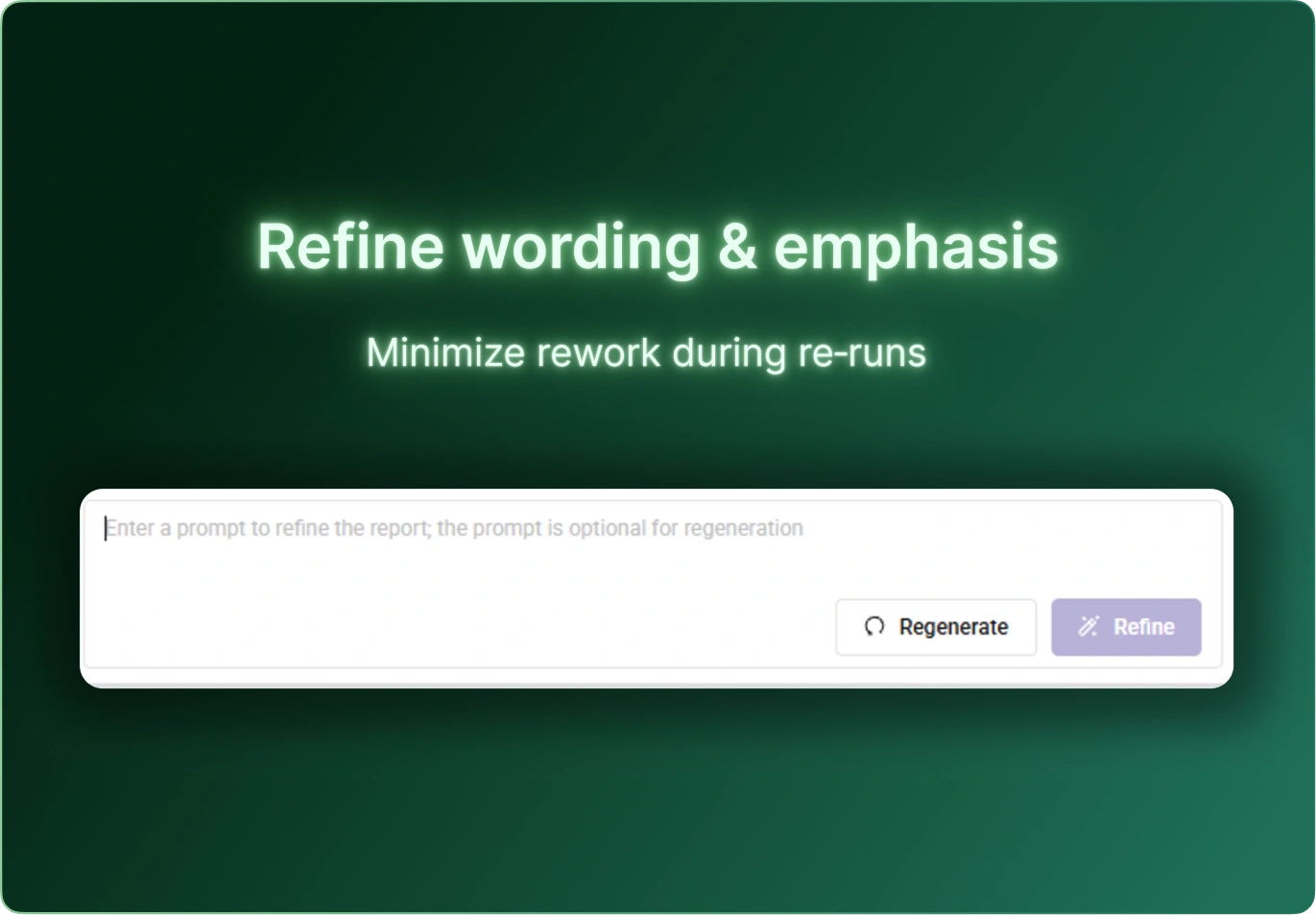
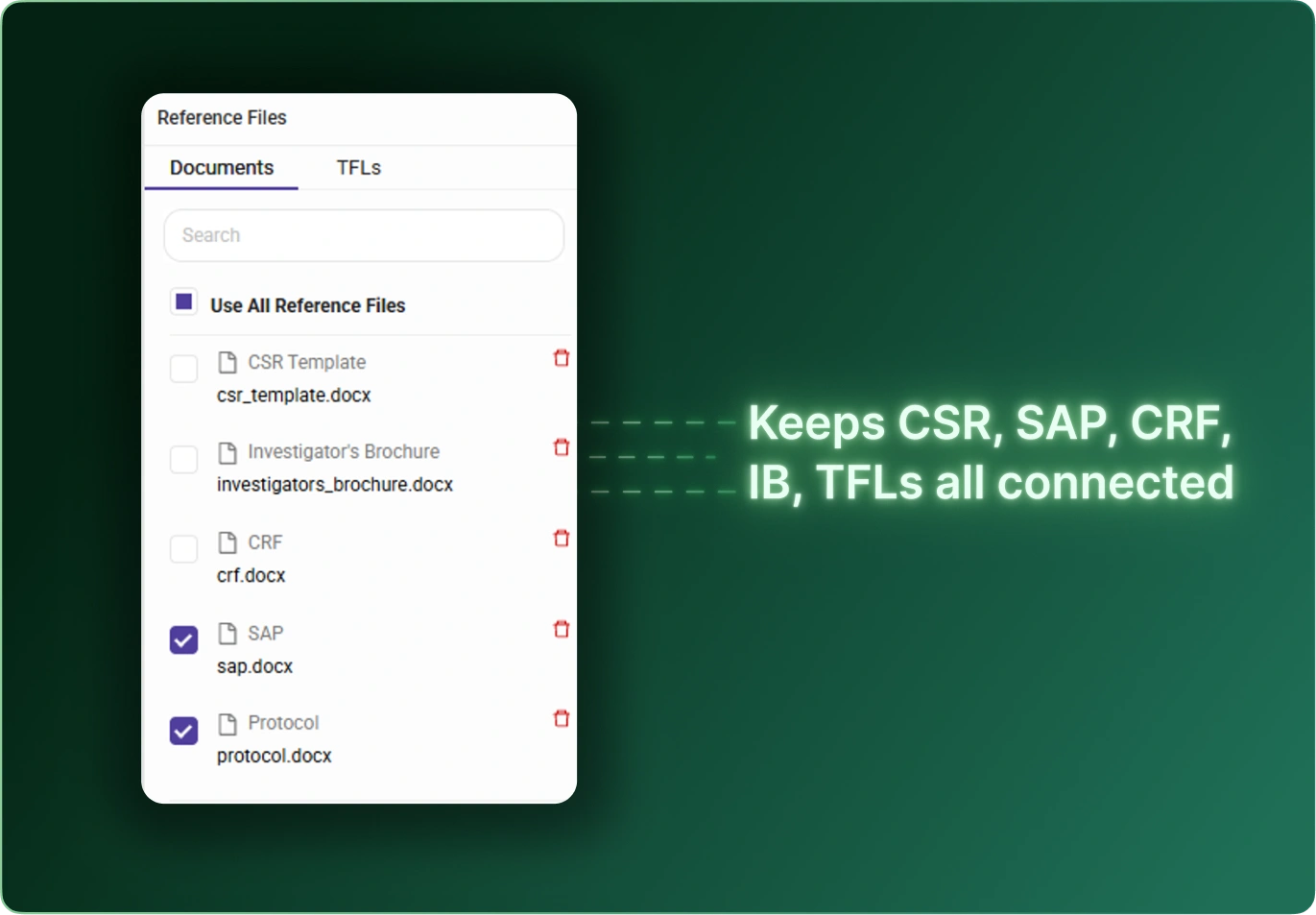
CSR Builder streamlines the creation of Clinical Study Reports by combining structured templates, automated content generation, and AI‑assisted drafting. Teams reduce manual writing time, improve consistency across studies, and accelerate delivery without compromising quality or compliance.