SAP Builder
Standardize and Automate Statistical Analysis Plans With Structured Metadata
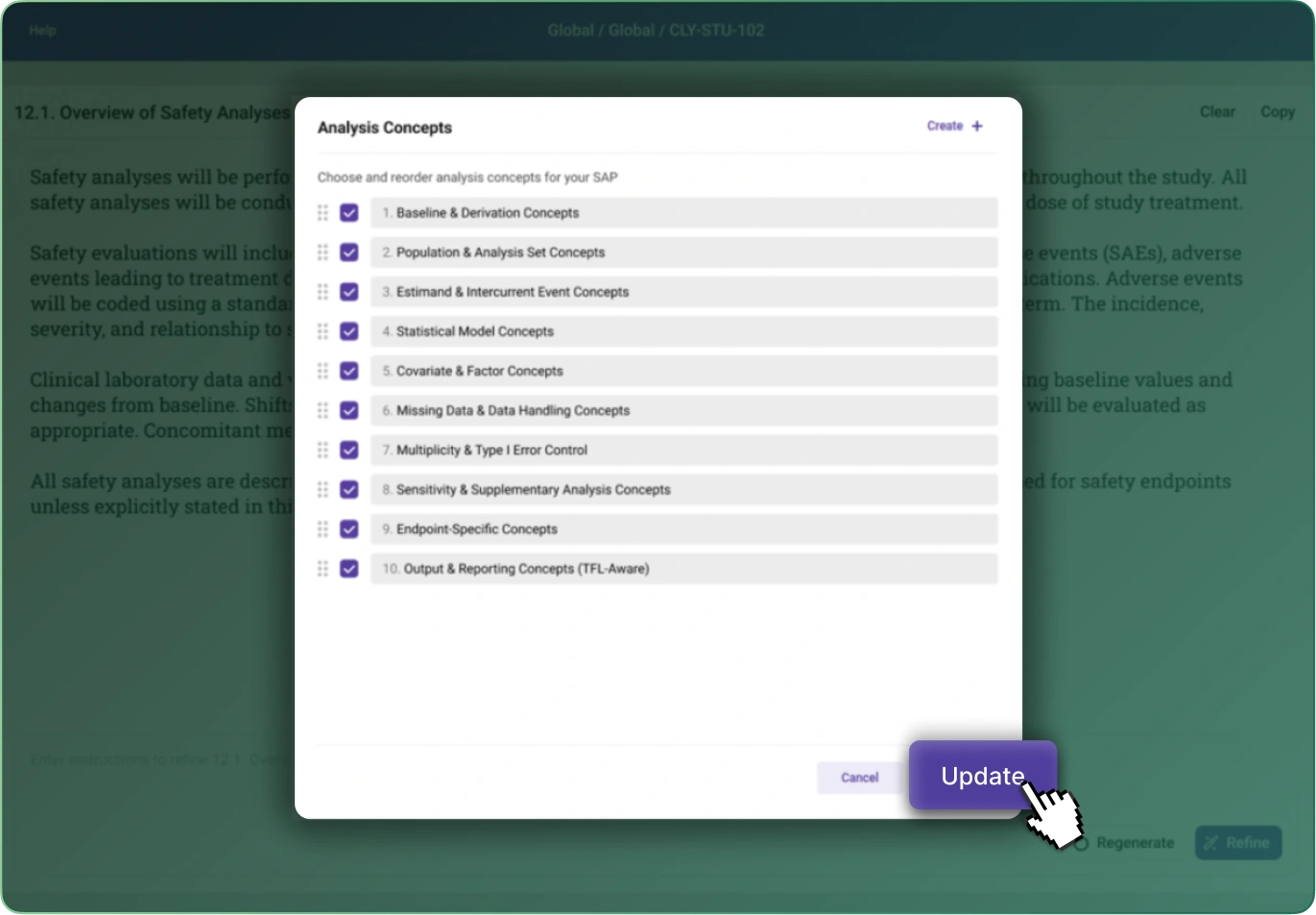
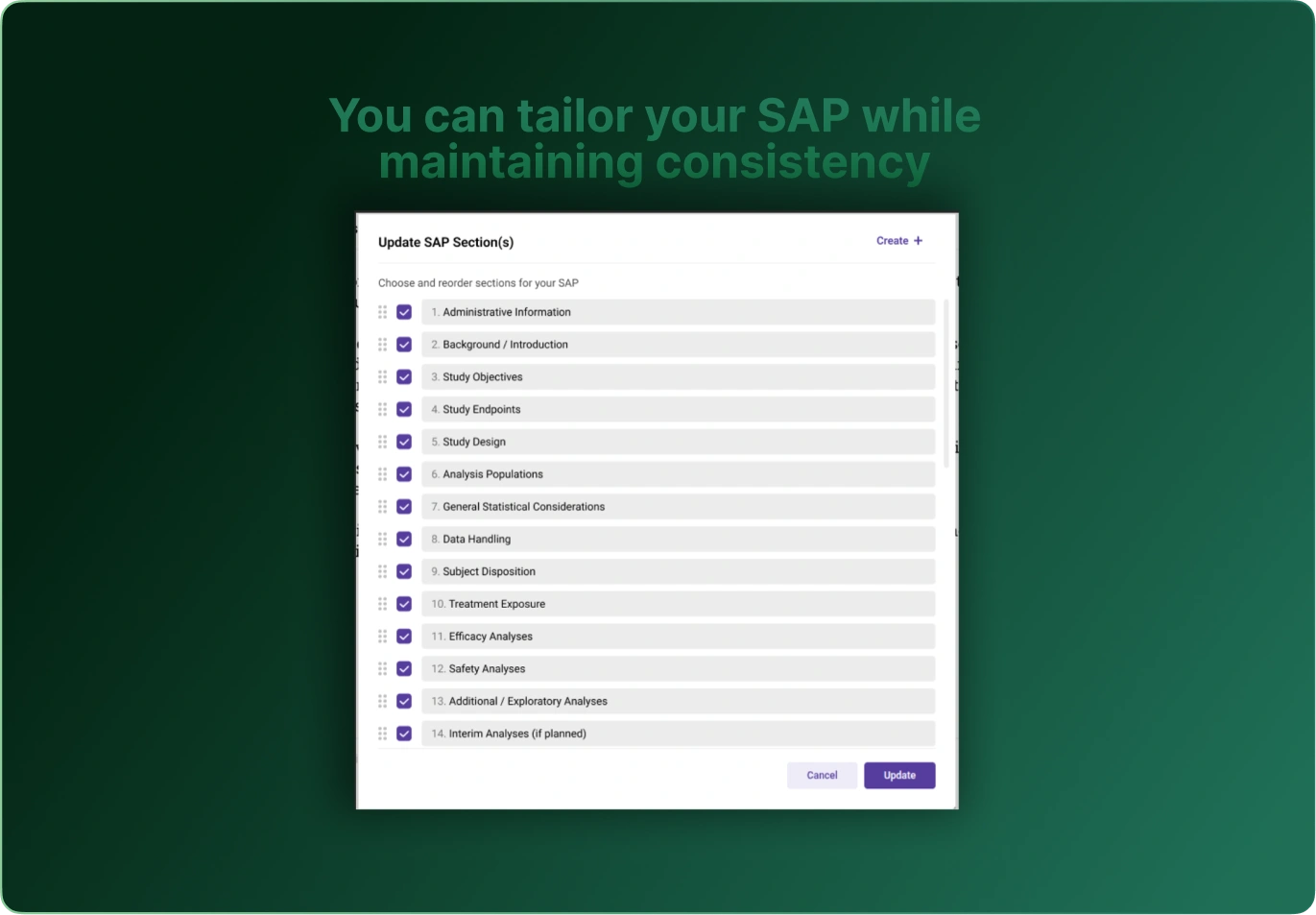
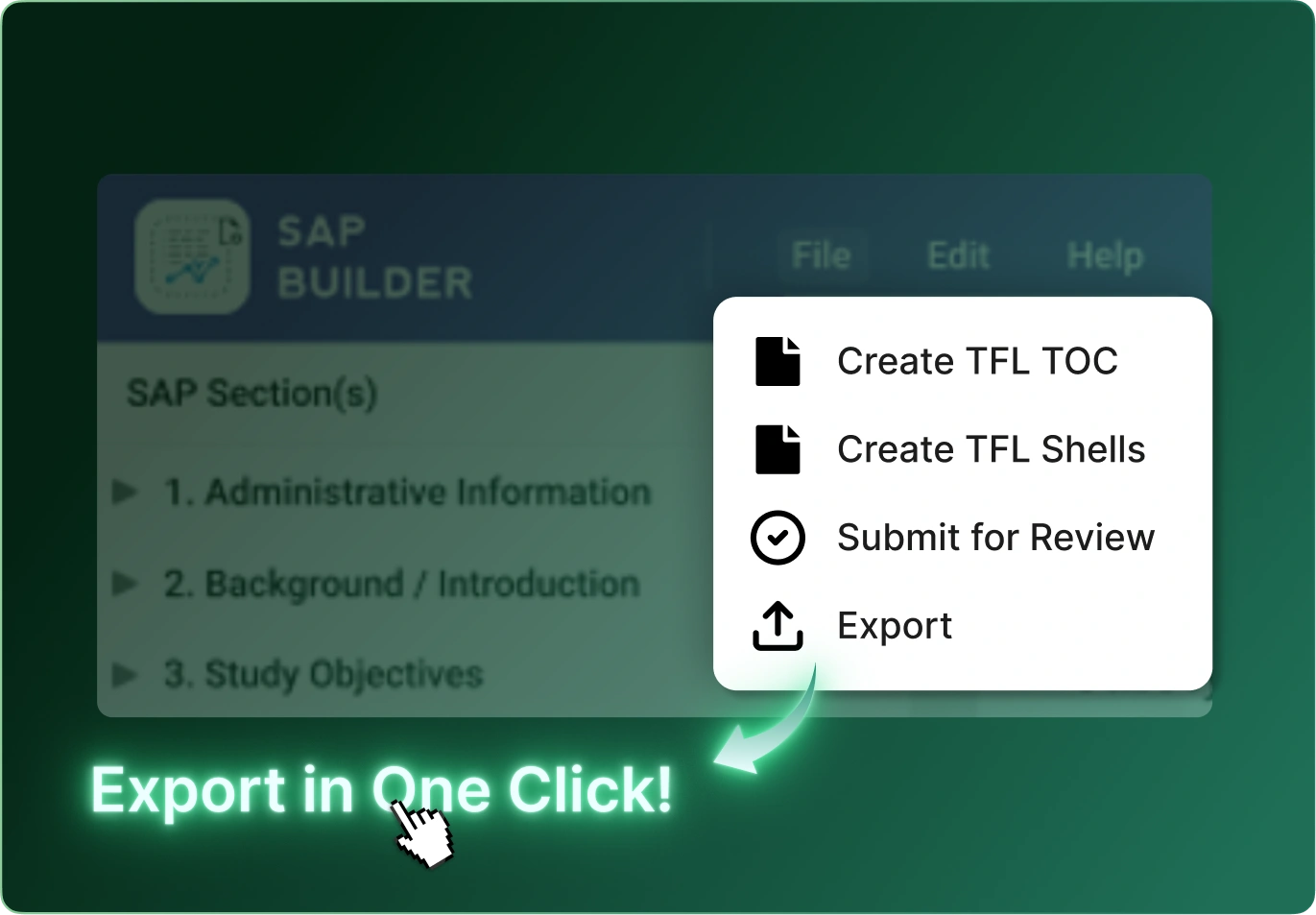
SAP Builder transforms the way teams create, manage, and govern Statistical Analysis Plans. By converting SAP requirements into structured, machine‑readable metadata, it eliminates ambiguity, accelerates downstream programming, and ensures traceability from protocol to final outputs.